A promising advance in the treatment of dry eye
11/03/2026
10/11/2022
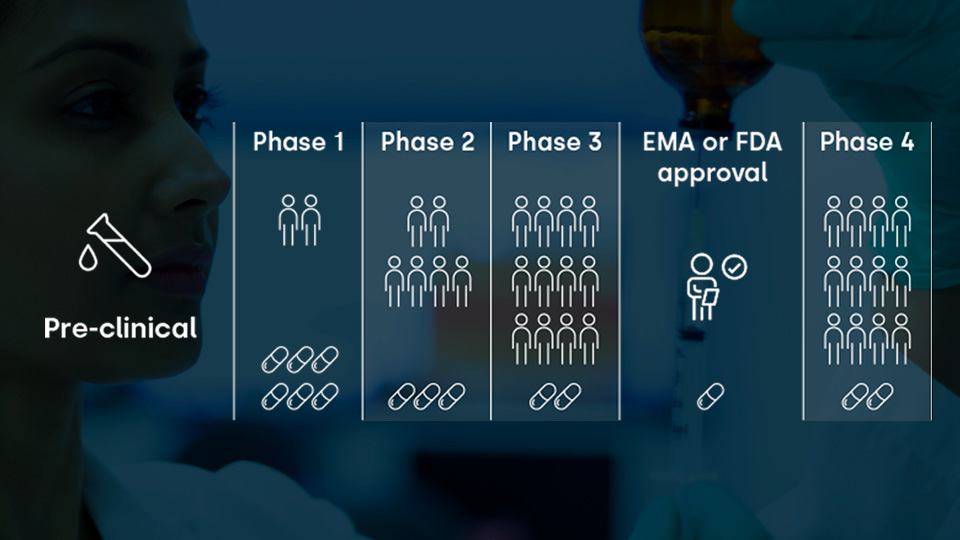
The drug development phases comprise various processes or stages that culminate in the performance of clinical trials. The trials themselves are also divided into different phases that range from the quest for the right dose, using just a few participants, to the efficiency of the new treatment among populations of thousands of people. At the same time, the safety and efficacy of the drug, the side effects and quality of life of participants in the study administering the new treatment are studied.
The different stages of a clinical study are sequential processes in which an attempt is made to demonstrate that a new drug is efficacious, effective, efficient, safe and improves the quality of life of patients.
Pre-clinical Phase
The drug is tested on animals before humans in order to check its efficacy and safety. If the necessary requirements are met at the end of this process, also known as the pre-clinical phase, the drug will then be tested on humans in a process called the clinical phase and is then divided into four phases:
Phase 1. Safety Evaluation
This phases enables us to understand how a drug acts on the body and how the body reacts to the drug (how it is absorbed, distributed and metabolised). This first stage is characterised by administering a new treatment or a new combination of drugs, for the first time to a healthy human. At the start of the phase, two low doses of the drug are used and then the dose is increased as long as the participant is not at risk. In some cases, the patients may stay in hospital for a few days for close monitoring.
Phase 2. Efficacy Evaluation
The main objective is to check if a new drug is efficacious in its new indication. The short-term adverse effects and safety risks associated with the researched drug are evaluated. The new treatment is administered for the first time to a participant with the targeted disease. That is to say, the drug has to show that it has a therapeutic effect on the disease for which it has been designed. Furthermore, it can also seek to be efficacious in drugs already on the market but for a new indication. In addition, dose ranges are also measured, from the lowest dose with an effect to the highest dose tolerated. In this phase there are more participants than in the previous phase and it usually lasts longer.
Phase 3. Confirmation of findings among a greater population of patients
The main objective of this third stage is to confirm that the researched drug has a therapeutic effect and that it is safe. If the results are positive, authorisation for the clinical use of the drug is requested. In the third phase, the number of participants and the duration of the trial increase to then confirm the data collected in phase 2. A more robust risk/benefit evaluation is carried out to determine whether the drug can be sold or not.
Phase 4. Review of treatment in clinical practice
These are the studies carried out once the drug is on the market and, therefore, a much larger population. They are designed to evaluate the long-term safety and efficacy of the drug. The main objective of this phase is to compile more data from much larger, non-homogenous populations, about safety and efficacy, new indications or economic matters. This enables us to find out, for example, if the drug will be valid for another type of disease (new indication) or, if the therapeutic effect (synergies) will become stronger in combination with another medication. But we will also be able to see in more detail whether or not it has serious consequences when a patients suffers from a different disease or is taking other medication at the same time (indications or contraindications).
Victòria Hernández Grima, Head of the Clinical Trials Department